CRISPR Remission™ Library
A CYNAERA framework for state-dependent, flare-aware gene editing across chronic and immune-volatile conditions
CRISPR-based therapies are advancing rapidly, yet remain limited by delivery constraints, immune variability, environmental influence, and the assumption of biological stability across patients. Current models focus on precision editing, but often fail to account for when, where, and under what conditions gene-based interventions can produce durable outcomes. CYNAERA’s CRISPR Remission™ Library offers one of the most advanced system-level frameworks in the world, designed to address instability, variability, and structural limitations in gene editing across complex and chronic disease.
Explore how CYNAERA’s AI engines, predictive algorithms, and state-dependent modeling systems are transforming how CRISPR is applied across conditions, integrating environmental burden, flare dynamics, viral persistence, and system stability to improve both safety and long-term effectiveness. CRISPR Remission™ is relevant to conditions affecting an estimated 1.5 billion people worldwide, spanning chronic, immune-volatile, genetic, and post-infectious disease systems.
Conference Recognition
CRISPR Remission™ was presented at CRISPRMED26, a leading international conference focused on genomic medicine, gene editing, and translational therapeutics. The abstract, “CRISPR Remission: A Flare-Aware Gene Editing Pathway Engine for Immune-Volatile Chronic Disease,” was reviewed within an international scientific environment spanning gene therapy, oncology, rare disease research, computational genomics, and pharmaceutical development.
Reviewers included researchers and scientific leaders from institutions across Europe, as well as industry representation from AstraZeneca, reflecting cross-sector evaluation of the concept’s relevance to real-world therapeutic development. This positioning underscores the growing recognition of immune volatility, patient heterogeneity, and flare dynamics as critical factors in the safe and effective deployment of gene editing technologies.
Where Gene Editing Has Already Changed the Remission Landscape
CRISPR Remission™ builds on a reality that is already unfolding across multiple areas of medicine. Gene editing is no longer limited to experimental correction of rare mutations. It is actively reshaping how remission is approached in complex disease, particularly where immune dysfunction, persistence, or recurrence have historically limited treatment success.
Approved Genetic Disease Applications
The clearest proof of real-world impact comes from genetic blood disorders. CRISPR-based therapies have now been approved for conditions such as sickle cell disease and transfusion-dependent beta thalassemia, where edited hematopoietic stem cells can restore functional hemoglobin production and eliminate the need for ongoing transfusion or reduce disease burden substantially (CRISPR Therapeutics, 2023; Frangoul et al., 2021).
These cases demonstrate that gene editing can move beyond symptom management to produce durable, system-level change in human disease. However, they are largely applied in biologically stable contexts, where intervention timing and immune volatility are less dominant constraints.
Cancer and Immune-Directed Therapies
In oncology, gene-edited and engineered immune cell therapies have already demonstrated the ability to induce deep remission in certain cancers, particularly hematologic malignancies. These approaches, including CRISPR-modified T cells and related platforms, show that targeted immune system reprogramming can eliminate disease at a systems level rather than suppress it temporarily (Stadtmauer et al., 2020; Nature Biotechnology, 2022). While outcomes vary across tumor types, the core insight is consistent: durable remission is achievable when the immune system itself becomes the therapeutic engine.
Autoimmune Disease and Immune Reset
The most important bridge to CRISPR Remission™ lies in autoimmune disease. Recent clinical work using engineered immune-cell therapies has shown early evidence of drug-free remission in conditions such as systemic lupus erythematosus and systemic sclerosis, where pathogenic B cells are selectively targeted and eliminated (Mackensen et al., 2022; Nature Medicine, 2025). These findings signal a shift from chronic suppression to functional immune reset. They also highlight a critical gap. Even in these advances, intervention timing, disease-state fluctuation, and environmental load are not yet fully integrated into deployment strategies.
Why This Matters for Infection-Associated Chronic Conditions
Together, these developments establish a clear trajectory. Gene editing is evolving from static correction toward dynamic immune modulation, with increasing evidence that remission is possible in complex, multi-system disease. Infection-associated chronic conditions, including ME/CFS, Long COVID, and related disorders, present a different challenge. These conditions are defined not only by immune dysfunction, but by instability, relapse patterns, environmental sensitivity, and fluctuating physiologic states that can alter treatment response over time. CRISPR Remission™ extends the remission paradigm into this domain by introducing a missing layer in current gene-editing models. Rather than assuming biological stability, it evaluates when intervention is most likely to hold, aligning gene-editing strategies with immune state, flare dynamics, and system-wide conditions that influence durability, safety, and long-term outcome.

CRISPR Remission™ Research Library
Next Generation Individual Gene Editing
-
Personalized CRISPR Remission™ for Long COVID
Diagnostic and Trial Infrastructure
Environmental and Flare Dynamics
-
SymCas™: Symptom Cascade Modeling

Why CYNAERA Chose CRISPR Remission™
CYNAERA’s CRISPR Remission™ work did not begin as an academic exercise. It began because complex chronic illness and genetic disease have shaped my life and my family in ways medicine has too often failed to prevent, recognize, or respond to in time. I will never forget, the moment my family got the news that my cousin died from complications of sickle cell disease before either of us ever made it out of elementary school.
I nearly died from Long COVID before age 40. My son developed Long COVID as a young child, and I have a nephew whose Long COVID continues to worsen even though he is still not out of high school. Another cousin became paralyzed from the waist down after mpox, also before age 40.
The pattern became impossible to ignore. I saw the genetic and systemic links, and I realized that generations of my family were at risk of a future shaped by severe disability, homebound life, and early loss if someone did not think differently and act earlier. That is why I intentionally set out to engineer my own treatment and cure. This was not an accident.
This library was built to make personalized CRISPR Remission™ legible in the real world, where disease is dynamic, not static, and where timing, environment, and system state can determine whether an intervention holds. This is a living intelligence structure built from survival, grief, and the belief that families deserve a more hopeful future.
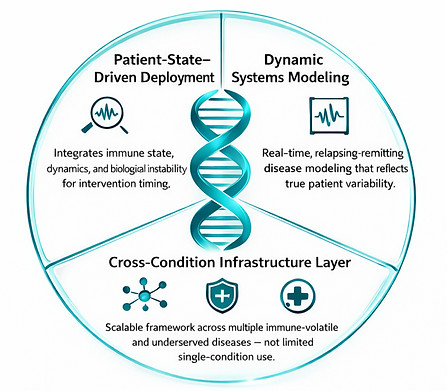
How CRISPR Remission™ Differs
CRISPR Remission™ introduces a fundamentally different approach to gene editing:
Patient-State–Driven Deployment
Integrates immune state, flare dynamics, and biological instability as core variables in determining when and how gene editing interventions are applied.
Dynamic Systems Modeling
Replaces static baseline assumptions with real-time, relapsing-remitting disease modeling that reflects true patient variability.
Cross-Condition Infrastructure Layer
Functions as a scalable framework across multiple immune-volatile and underserved diseases rather than being limited to single-condition CRISPR applications.
Applications in Therapeutic Development and Research
CRISPR Remission™ is designed as a strategic infrastructure layer for organizations developing, evaluating, or operationalizing gene editing in complex disease environments. Its commercial value lies in helping therapeutic developers, research institutions, platform companies, and translational partners address a major gap in current genomic medicine: the lack of deployment frameworks built for biologically unstable patient populations.
The framework is relevant across gene therapy and CRISPR-based therapeutic development, particularly where treatment success may be influenced by fluctuating immune activity, variable disease state, relapse patterns, or environmental sensitivity. In these settings, individualize CRISPR Remission™ can support a more informed development strategy by introducing timing logic, stability assessment, and patient-state awareness into therapeutic planning. This creates potential value not only in scientific design, but also in risk reduction, development efficiency, and strategic positioning.
CRISPR Remission™ also has direct application in clinical trial design and protocol optimization. For sponsors, trial designers, and translational teams, it offers a way to better account for disease volatility that may otherwise distort enrollment assumptions, subgroup analysis, intervention timing, and outcome interpretation. This is especially relevant in conditions where standard trial models have underperformed because they were built around static rather than fluctuating patient profiles.
In patient stratification and eligibility modeling, the framework supports more precise identification of intervention-ready subgroups by incorporating biological instability, flare probability, and disease-state variation into selection logic. This has potential utility for companies seeking to improve targeting, reduce avoidable variability, and build more realistic pathways toward therapeutic application in underserved or difficult-to-model populations.
CRISPR Remission™ in Autoimmune and Immune-Mediated Disease
CRISPR Remission™ is increasingly relevant across autoimmune disease, where durable remission depends not only on targeted immune-cell modification, but on system state, inflammatory load, and biologic timing. As stated in CYNAERA’s work on lupus and type 1 diabetes, CRISPR-based strategies show the strongest potential when aligned with immune stability and disease-specific volatility rather than applied uniformly across patients.
The Eve Research Project adds a real-world pattern layer to this model by capturing overlooked autoimmune signals across women, families, hormonal shifts, flare behavior, and lived disease progression before those patterns are consistently recognized in formal research or care systems.
This framework extends across a broader spectrum of autoimmune disease, including endocrine, systemic, and glandular conditions. Emerging applications include Hashimoto’s thyroiditis and Sjögren’s syndrome, where immune-driven tissue damage, hormonal interaction, and environmental factors contribute to disease persistence and variability. Additional autoimmune conditions, including rheumatoid arthritis, multiple sclerosis, systemic sclerosis, and immune thrombocytopenic disorders, represent key areas for continued expansion within a state-dependent CRISPR Remission™ model.
Platform Integration and Commercial Value
CRISPR Remission™ is also well positioned for integration into digital twin systems, simulation platforms, and advanced therapeutic modeling environments. In this context, it can strengthen platform capability by adding a layer of state-responsive logic that reflects real-world patient variability rather than idealized baseline assumptions. This makes it relevant to organizations building next-generation infrastructure for predictive modeling, translational simulation, and therapy optimization.
More broadly, the framework has value for translational research groups, biotech partners, and strategic collaborators working in relapsing-remitting, immune-volatile, and historically neglected disease categories, including conditions with limited or no FDA-approved treatment options. By reframing instability as a design variable rather than a disqualifying complication, customized CRISPR Remission™ supports a more scalable and commercially meaningful approach to therapeutic development in populations that have often been excluded from conventional innovation pathways.
From a business perspective, CRISPR Remission™ is suited for licensing, partnership, co-development, and integration into existing genomic medicine pipelines. Its utility extends across therapeutic strategy, protocol architecture, simulation, and platform enhancement, making it relevant not only as a scientific concept, but as an enterprise asset with cross-condition and cross-sector application. Based on CYNAERA modeling, the framework has the potential to reduce off-target risk by 50 to 70 percent, reduce toxicity by 30 to 50 percent, accelerate trial timelines by 6 to 12 months, and lower projected per-patient costs to under $500,000 compared with current therapeutic pathways that may exceed $1.5 million. These gains are tied not only to scientific optimization, but to reduced adverse events, broader eligibility, more efficient trials, and expanded access across complex disease populations.
What Is CRISPR²™?
CRISPR²™ is CYNAERA’s next-generation, flare-aware gene editing system. It expands beyond target precision alone by integrating timing, biological readiness, environmental load, and recovery dynamics into how gene editing is deployed. In complex, immune-volatile disease, CRISPR²™ helps determine not just what to edit, but when intervention is most likely to be safe, stable, and durable.
CYNAERA Gene Editing Bundles
CRISPR Remission Core™
Foundational framework for flare-aware gene editing deployment, including pathway logic, immune volatility modeling, and translational positioning for therapeutic pipelines.
Flare-State Readiness Bundle™
Integrates SymCas™, VitalGuard™, and CYNAERA pathway logic to identify stability windows, environmental risk load, and relapse probability prior to intervention.
Immune-Volatile Translation Bundle™
Advanced package for institutions and companies working across Long COVID, Lyme, ME/CFS, autoimmune and post-viral conditions, supporting cross-condition deployment strategy.
